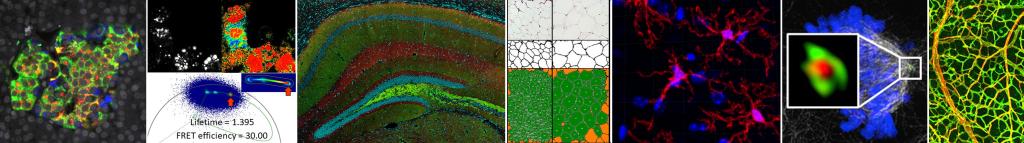
The Microscopy and Image Analysis Core (MIAC) provides state-of-the-art microscopy and image analysis software to enhance diabetes-related research at the University of Michigan that would otherwise be beyond the scope and resources of most investigators. The MIAC provides expertise, advice, training, and access to equipment for microscopic and morphometric analysis of both protein and RNA in biologic samples in support of research focused on diabetes and its complications or related endocrine and metabolic disorders. The MIAC also provides expertise in protein targeted antibody staining for microscopy, live cell imaging and use of fluorescent probes for study of cell signaling.
The primary goal of the MIAC is to provide MDRC researchers with expert consultation, training and support for routine and advanced microscopy and image analysis techniques. Each project begins with a consultation between the investigator and either Drs. Antonetti or Lentz to discuss the investigator’s needs and objectives.
The MIAC is supported with institutional funds.
How We Serve Your Research Needs
Consultation and Advice on Microscopy and Immunohistochemistry
-
MDRC investigators receive consultation prior or during training
-
Advice focuses on immunological staining of fixed cells/tissues, fluorescent-based dyes for live cell imaging, microscopy techniques, processing and visualization of microscopic images, and morphometric features analysis
-
Core Director and Laboratory Director typically handle the consultations
-
Recommendations to other experts from the UM community provided when needed
-
Investigators can use Core equipment at an hourly rate or obtain services by core personnel for additional fees
Confocal Microscopy
-
The core has a Nikon A1, a Leica Stellaris 8, and a Leica TCS SP5 Confocal Microscope
-
Each system has its strengths to optimize acquisition for a variety of imaging configurations
-
Investigators are guided to the confocal system that fits best their needs
-
Images can be analyzed by users after training or Core personnel
-
Users receive four free hours of instruction
Image Analysis
-
The service includes morphometric image analysis and preparation of publication-quality images
-
The Core has software programs like AutoQuant and Leica's Lightning for image restoration
-
Quantitative analysis of images can be done using MetaMorph, ImageJ, Aivia or MATLAB software
-
3D reconstruction and fluorescence intensity quantification can be performed with Imaris and Aivia software
-
Publication-quality figures and montages preparation available with Photoshop, Illustrator, Prism, and Powerpoint software
-
Instruction in the use of software provided by Laboratory Director
Widefield Light Microscopy
-
An inverted Nikon TiE microscope is available for imaging and standard fluorescent signal acquisition (blue, green red and far-red) with a sensitive Photometrics CMOS camera
-
Equipped with specialized excitation and emission filters to allow users to capture fluorescence-based images of transfected green fluorescent protein (GFP) chimeric molecules, eCFP and eYFP signals for Förster Resonance Energy Transfer (FRET)
-
Image acquisition are done with Nikon Elements software
Live Cell Microscopy
-
Consultation begins with the Laboratory Director to decide appropriate microscopy technique between confocal and wide-field fluorescence microscopy
-
Stage-top devices available for temperature and CO2 regulation
-
Support a variety of dyes and probes for sensing, labeling, and tracking
-
The core has experience to date using fura-2, fluo3, calcium green and Magfura dyes for Ca2+, SBFI for Na+, BCECF for H+, SPQ for Cl-, cSNARF-1 AM for pH, NADH/NAD+ sensors, lucifer yellow and lissarhodamine as gap junction tracers, and eCFP, eGFP, eYFP, DSRed, mCherry fluorescent proteins as markers for transfected cells, membrane potential sensors (TMRM, TMRE, JC-1), labeling mitochondria (MitroTracker and Bacmam MitoGFP/RFP) and following endocytosis
-
Frequency of image collection can be optimized to manage data files size and minimize probe bleaching
Specialized Techniques
-
Consultation and training available for complex imaging techniques like FRET, FRAP, FLIP, FLIM, photoactivation and uncaging
-
Nikon A1 and Leica Stellaris 8 systems have specialized protocols for image acquisition
-
Instrumentation and training for FRET is available but FRET is not offered as a routine service since it requires a large developmental component
Key Personnel
David A. Antonetti, PhD
Core Director
734-232-8230
dantonet@umich.edu
Stephen I. Lentz, PhD
Laboratory Director
734-647-8233
lentzs@umich.edu
Contact Us
Microscopy and Image Analysis Core Laboratory
Brehm Tower, Room 6245
1000 Wall Street
Ann Arbor, MI 48105-5714
734-647-8233
lentzs@umich.edu
